Other chemical compounds have been used as popular fertilizers over the last century. Ammonium nitrate (N2H4O3.) is one such compound and has an NPK rating of 34-0-0. Urea, on the other hand, has an NPK grade of 46-0-0, making it more economical to transport. Ninety percent of synthetic urea produced now is for fertilizers.
 2. IMPURITIES AND IMPROPER USE OF UREA FERTILIZERS CAN DAMAGE PLANTS
A common impurity in urea fertilizers is biuret (C2H5N3O2), which can be broken-down in soil but does so
over a long period of time, and is phytotoxic during the process.
2. IMPURITIES AND IMPROPER USE OF UREA FERTILIZERS CAN DAMAGE PLANTS
A common impurity in urea fertilizers is biuret (C2H5N3O2), which can be broken-down in soil but does so
over a long period of time, and is phytotoxic during the process.
As with any source of nitrogen, urea itself can damage plants: nitrogen impairs or completely impedes seed germination, and too much nitrogen can give crops a “burn.”

3. EXISTING SOIL BACTERIA BREAKS DOWN UREA FERTILIZERS The first step in making the nitrogen in urea (CH4N2O) available to plants is by converting it to either ammonia (NH3) or ammonium ions (NH4+) and bicarbonate ions (HCO3−). Naturally-occurring soil bacteria—broadly-called ammonia-oxidizing bacteria (AOB)—accomplish this rapidly with the enzyme urease.
Thereafter, through a process called nitrification, ammonia is oxidized to nitrite. Nitrite is oxidized to nitrate by nitrite-oxidizing bacteria (NOB).
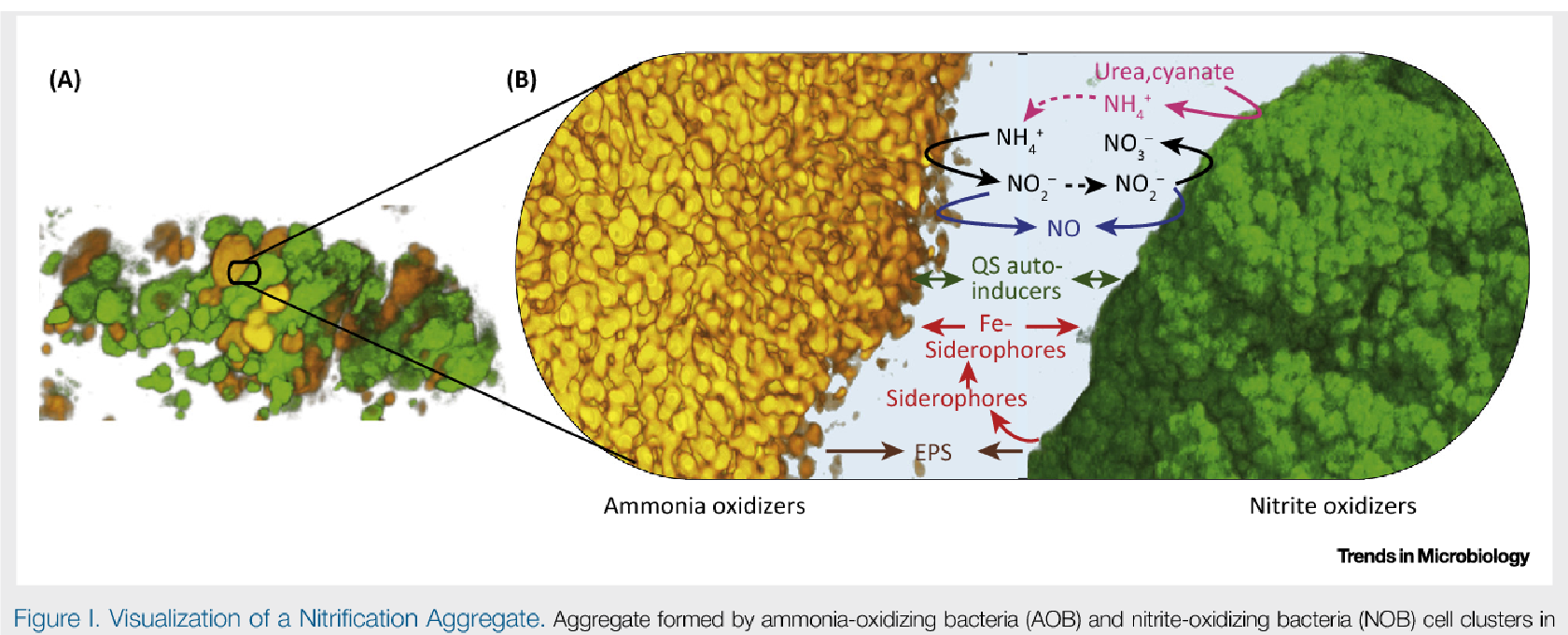 It is both ammonium and nitrate which are the forms of nitrogen that are most readily absorbed by plants.
4. UREA FERTILIZERS IMPACT SOIL PH
During the nitrification process, there are increases in the number of free hydrogen ions (H+) in the soil,
which creates acidity. Additionally, when plants take up ammonium ions (NH4+), they also release hydrogen
ions into the soil.
It is both ammonium and nitrate which are the forms of nitrogen that are most readily absorbed by plants.
4. UREA FERTILIZERS IMPACT SOIL PH
During the nitrification process, there are increases in the number of free hydrogen ions (H+) in the soil,
which creates acidity. Additionally, when plants take up ammonium ions (NH4+), they also release hydrogen
ions into the soil.
Thus care must be taken to neutralize excess acidity, by using compounds like Effective Calcium Carbonate (ECC).
 5. HEALTHY PLANT GROWTH DEPENDS ON QUALITY, NOT QUANTITY
As mentioned above, too much urea and by extension too much nitrogen can have a negative impact on plants.
It can also have a negative impact on surrounding environments: nitrate is very mobile in water, and
leaching from agriculture is harmful to waterways.
5. HEALTHY PLANT GROWTH DEPENDS ON QUALITY, NOT QUANTITY
As mentioned above, too much urea and by extension too much nitrogen can have a negative impact on plants.
It can also have a negative impact on surrounding environments: nitrate is very mobile in water, and
leaching from agriculture is harmful to waterways.
Thus while urea fertilizers are on the whole a climate-smart and economical choice, care must be taken in their application in order to provide the maximum benefit to your crops with a minimal impact on the environment.
Using i-PlantNutrition’s tools can help in making those choices. In fact, in our software there is a specific function for pre-planting fertilization where the different forms of nitrogen (Urea, Ammoinium and Nitrate) are taken into account in order to reduce losses and leaching. This allows the User to not only save money and avoid damages to the environment, but also to deploy the nutrient more effectively to the crop.





